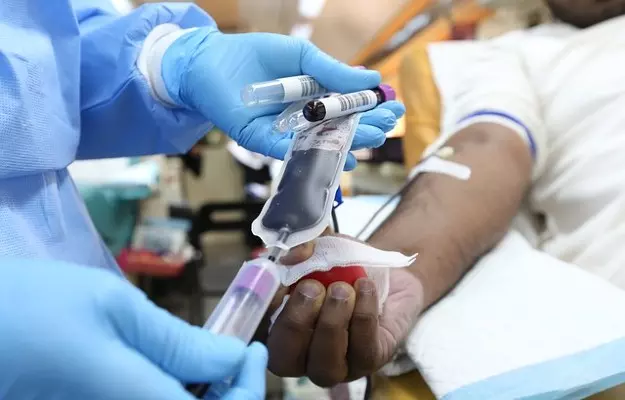
Blood banks around the world depend on three types of donation of blood and blood products: voluntary unpaid donations, replacement blood (for example, you may donate blood if an acquaintance who is in hospital needs it), and paid donations.
These donations have suffered over the last few months. Not only has an extended lockdown in the country since 25 March restricted the movement of people, but many medical services around the country have also had to be curtailed during the COVID-19 pandemic. One of the fallouts of this is a growing shortage of blood and blood components in blood banks.
Reports of thalassemia patients being unable to avail blood transfusion at a Lucknow hospital, and news from other parts of the country, have highlighted this rising concern amid the COVID-19 crisis, which has crippled many departments within the medical world. Despite assurances from the Ministry of Health and Family Welfare that hospitals continue blood transfusion and blood donation drives, the shortage has been amplified by the national lockdown which was extended for a third time in the month of May.
Read more: 12 things people want to know about thalassemia
According to the World Health Organization (WHO), "if 1% to 3% of a country's population donates blood, it would be sufficient for the country's needs". India does not meet even the lower limit of 1% of this recommendation, falling short by 1.9 million units per year in 2018.
The National Blood Transfusion Council on 25 March - the first day of the national lockdown - had issued a list of guidelines for the continuation of blood transfusion services in the country to maintain the supply of blood samples for critically ill patients, as well as for patients who depend on blood transfusions as a life-saving measure, like people with thalassemia (a blood disorder).