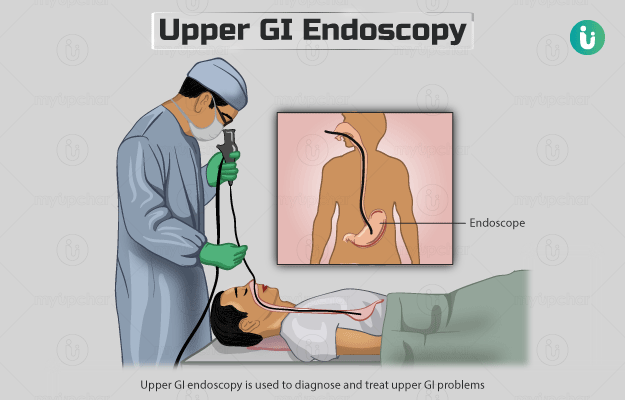
What is a Carcinoembryonic Antigen (CEA) test?
Carcinoembryonic Antigen or CEA is a protein that is present abundantly in the tissues of a developing foetus. However, its levels reduce or become undetectable after birth. Healthy adults have very little to no CEA circulating in their bloodstream, but CEA levels may start rising in certain types of cancer.
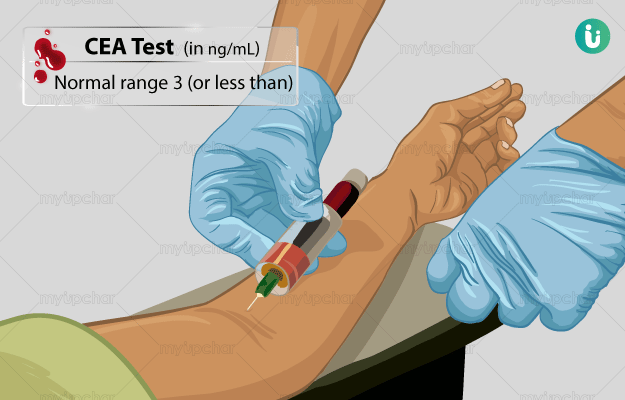
CEA test uses Carcinoembryonic Antigen (CEA) as a tumour marker for detecting certain kinds of cancer. It is also an important tool for evaluating a person's response to cancer therapy.
It was initially used in diagnosing only colon cancer (cancer of the intestine). However, eventually, the levels of CEA were found to increase in other types of cancer too such as rectal, ovarian, lung, thyroid and liver.