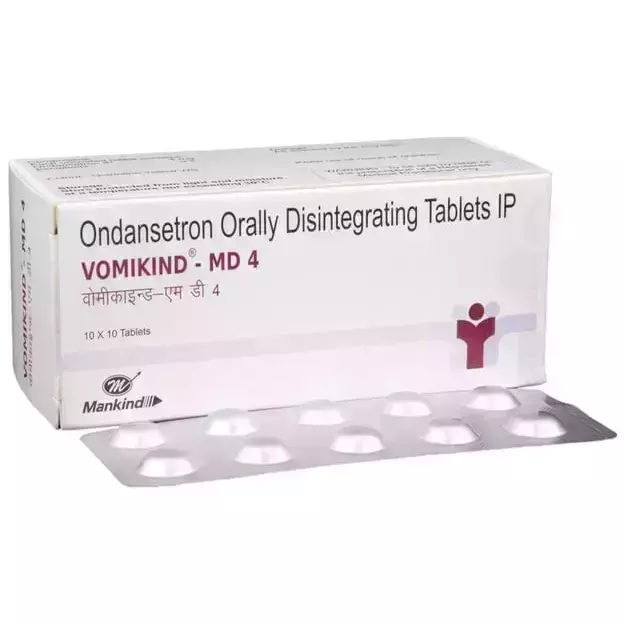
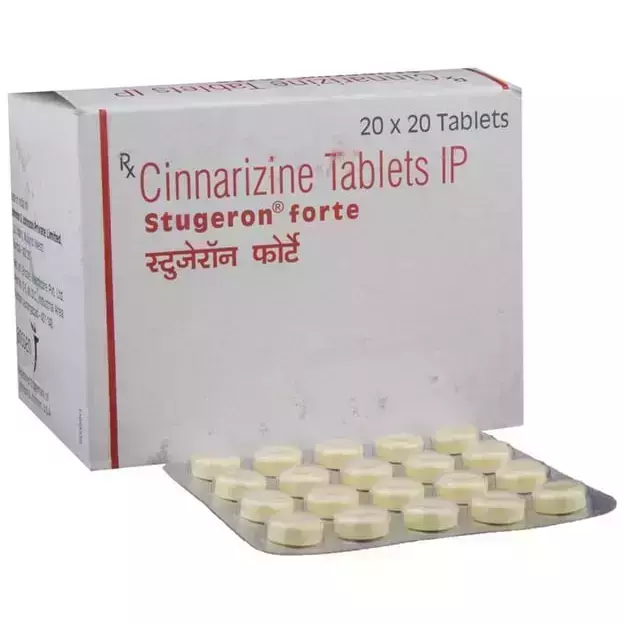
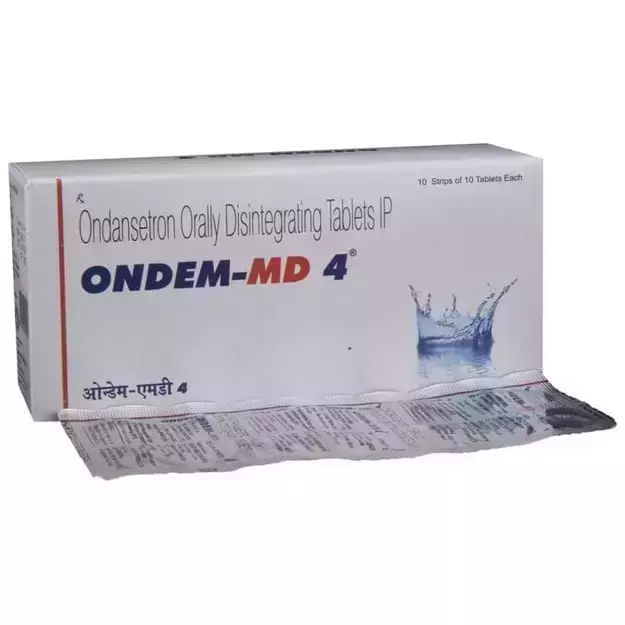
Bumper Offer - Urjas oil Just @ Rs. 1
X

- हिं - हिंदी
- En - English
- Consult
- Health Concerns
-
- Skin Issues
- Acne
- Fungal Infection
- Weight Management
- GainMan
- Weight Loss
-
- Hair Problems
- Hair Growth
- Hair Dandruff
- Self-Analysis
-
- Chronic Diseases
- Diabetes
- Heart Care
- Sleep Support
- Liver Care
- Stress & Anxiety
- Our Brands
- Medicine A-Z
-
Health A-Z
-
- Treatments
- Home Remedies
- Herbs
- Surgery
- Lab Test
- Therapy
- First Aid
- Ayurveda
- Homeopathy
-
- Yoga And Fitness
- Fitness
- Yoga
- Weight Loss
- Weight Gain
-
- Other Topics
- Baby Names
- Beauty
- Healthy Foods
- Tips
- Health News
- Pet Health
- Men Health
- Medical Cannabis
- Login / Sign Up