COVID-19 is an infection caused by SARS-CoV-2, a new coronavirus that was unknown until December 2019. As of 27 July 2020, there were more than 1.6 crore cases of this new coronavirus infection globally and the death toll had crossed 6.4 lakh. (Read more: How SARS-CoV-2 spreads)
Doctors and scientists - in their urgent search for a cure or a preventive measure to stop the spread of this infectious disease - have turned their attention to the patients who have recovered from the SARS-CoV-2 infection without any complications. (More than 1.7 lakh people had recovered from the illness, as of end-March 2020.)
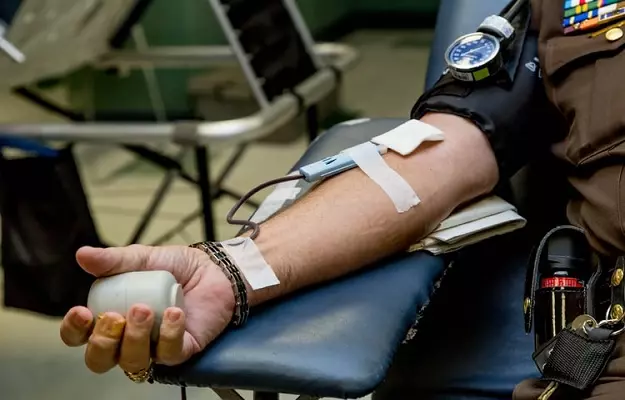
The reason: the immune systems of the patients who have recovered have produced antibodies against the virus. These antibodies neutralize the virus and prevent it from infecting the healthy cells. Doctors have found a way to transfer these antibodies into other people - to both prevent and treat the infection: this transfer of ready-made antibodies against the virus is known as convalescent plasma therapy or passive antibody therapy.
Difference between vaccines and convalescent plasma therapy (passive antibody treatment)
Our immune system has the ability to detect any foreign object that enters our body. That foreign object is called an antigen. To fight that antigen, the body forms a fighting agent which is called an antibody. Most of us have got vaccinations in childhood to protect us from specific diseases. These vaccinations are called active vaccinations as they do not directly provide immunity to our body - instead, they activate our immune system to form antibodies against the disease.
However, in convalescent plasma therapy (passive antibody therapy), the specific antibodies required to kill a specific agent are directly delivered into the body. Plasma therapy or passive antibody administration is one of the most potent ways of providing immediate immunity to the people who are susceptible to a specific disease. Plasma therapy has previously been used for viral diseases such as poliomyelitis, measles, mumps, and influenza in the 1920s.