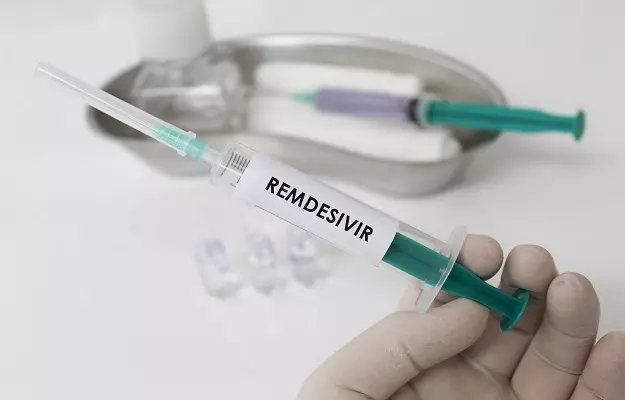
Remdesivir is still under investigations to study its efficacy and side effects (if any).
In vitro (lab) and in vivo (animal) studies show that Remdesivir is effective against SARS-CoV the causative agent of SARS (Severe Acute Respiratory Syndrome) and MERS-CoV, the causative agent of MERS (Middle Eastern Respiratory Syndrome).
At least four different Phase 3 trials are underway (or are almost done) to test the effect of Remdesivir on COVID-19 patients. Phase 3 clinical trials involve re-testing the findings of the earlier trials, with more than 1,000 patients, to study the safety and efficacy of a drug.
Three of these trials are being funded by US-based agencies - one by the NIH's National Institute of Allergy and Infectious Diseases (NIAID), and two at Gilead Sciences. The fourth one is being conducted at INSERM (Institut National de la Santé et de la Recherche Médicale) in France.
The National Institute of Health (NIH) started conducting clinical trials on Remdesivir in late February. The study, which includes more than 1,000 patients, spread across 68 sites in the USA, Europe and Asia, was till then the biggest study on Remdesivir. Findings from the trial (mentioned in an April 29 news release by the NIH) indicated that:
- Remdesivir is more effective than placebo for improving lung function in COVID-19 patients.
- Preliminary data from the study showed that about 31% patients who received Remdesivir recovered more quickly than those who got a placebo.
- The mortality rate in Remdesivir group was 8% in comparison to the 11.6% in the placebo group, according to the release.
- Patients who got treated with Remdesivir were able to leave the hospital in 11 days in comparison to the placebo group who took 15 days to get discharged.
Out of the two Gilead trials initiated in March, one is for severe COVID-19 while the other is for moderate COVID-19. Between the two trials, the drug will be tested on more than 7,500 people.
The trials are still underway, as per the registry of clinical trials by NIH. However, Gilead has released (on its website) the results from the trials so far. As per the news release, the results show that:
- With a five-day or 10-day treatment plan with Remdesivir, patients showed clinical improvement by Day 10 and Day 11, respectively.
- More than 50% of patients were able to leave the hospital by Day 14.
Two other trials that began in China as early as February 2020 to study the effect of Remdesivir on mild, moderate and serious cases of COVID-19 were terminated due to lack of patients.
Read more: Ebola drug Remdesivir enters human trials for COVID-19
A recent study published in The Lancet showed the findings of a Remdesivir study done across various hospitals in China. As per the article, 237 patients were given Remdesivir versus a placebo intravenously in a 2:1 ratio. The first dose was 200 mg and after that, a 100 mg intravenous infusion was given from Day 2 to Day 10 once a day. Although the Remdesivir group noted faster improvement in symptoms in the patients who started treatment earlier, the drug did not seem to have any statistically significant effects on the treatment.
However, it is important to note that the Remdesivir group had more patients with heart conditions. Diabetes, heart diseases and hypertension were the most common comorbidity in all patients.