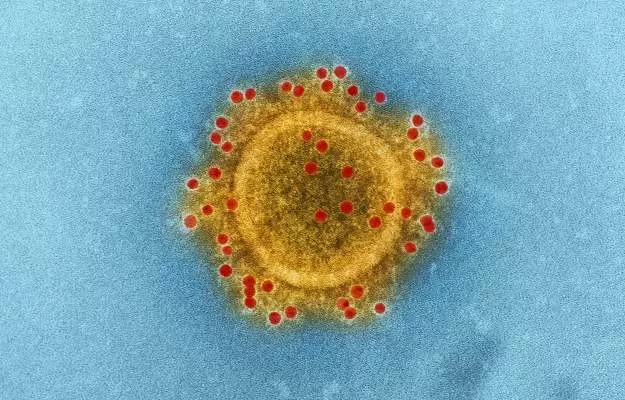
You would have noticed that when you get hurt, a bump appears in the affected area: there’s a bit of redness and swelling, and the area may even become a little hot to the touch. This is the most obvious kind of inflammation in the body.
Inflammation is a key tool that our immune system uses to heal the body after an injury, trauma or infection. Normally, an intricate system of proteins turns on and turns off this inflammation.
In some cases, however, the immune system mounts what scientists call an exuberant immune response. This means that while the sudden and localised immune response was necessary initially, it went overboard or became dysregulated at some point.
When this happens due to excessive production of cytokines—proteins that our immune cells use to talk to each other—it is known as a cytokine storm or cytokine storm syndrome (CSS). A cytokine storm is dangerous because it can trigger organ damage and failure and even death.
Though the term cytokine storm has gained currency lately in the context of viral infections, medical practitioners have known about some types of systemic immune system dysregulation for thousands of years: ever since people were first diagnosed with sepsis. (A cytokine storm often leads to sepsis but they are not the same thing).
That said, the term cytokine storm itself seems to have been coined in 1993 in relation to graft versus host disease, a complication of organ transplants. In 2003, it became linked to viral infections. Some of the viral infections that have been known to cause a cytokine storm in patients are:
- COVID-19
- H5N1 avian flu
- Severe acute respiratory syndrome or SARS
- Dengue
- Influenza
- Cytomegalovirus infection
- Epstein-Barr syndrome
Some non-communicable diseases that can trigger a cytokine storm include pancreatitis (inflammation of the pancreas, an organ that is responsible for the production of several digestive enzymes and hormones like insulin) and multiple sclerosis (an autoimmune disease in which the immune system attacks a protective sheath or layer around the nerves). Over the last 15 years, excessive cytokine production (cytokine release syndrome) has also been studied extensively in the context of cancer immunotherapies like CAR-T as a potential side-effect.
As the word storm indicates, it becomes more intense as time passes. A cytokine storm is best controlled in the initial stages before it causes end-stage damage to organs like the lungs, heart and kidneys.
This is, of course, easier said than done: the immune cells use a complex and overlapping system of cytokines to talk to each other through a process known as cell signalling. There are indeed different types of cytokines: some are proinflammatory (they cause inflammation), there are others that are anti-inflammatory. Still others, like interleukin-6 or IL-6, are pleiotropic—they can be pro or anti-inflammation.
Here’s what you need to know about what are cytokines, what is a cytokine storm, types of cytokines, the role of cytokines in respiratory infections like COVID-19, some signs of cytokine storm in patients and what to expect in a cytokine storm.